Does chlorobenzene give a nucleophilic substitution reaction?
However, in some adverse conditions and reagents, chlorobenzene can give nucleophilic substitution reactions. – Chlorobenzene when reacted with fused NaOH gives phenol. The reaction takes place at a high temperature. Phenol then again reacts with NaOH to give its salt.
What is the correct order of nucleophilic substitution in chlorobenzene?
Hence correct order is: Chlorobenzene < 4-Nitrochlorobenzene < 2,4-Dinitrochlorobenzene < 2,4,6-Trinitrochlorobenzene. Was this answer helpful? Arrange the following in order of increasing ease towards nucleophilic substitution.
What is the electrophilic substitution reaction of chlorobenzene?
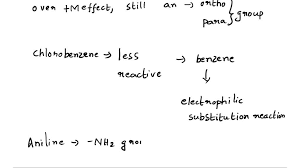
In electrophilic aromatic substitution reaction of chlorobenzene, the ortho/para ability of chlorine is due to its positive resonance effect (+R). The lone pair of electron on chlorine is involved in delocalisation with benzene ring and increase the electron density at ortho and para positions.
Does chlorobenzene undergo sn1 reaction?
Answer and Explanation: Chlorobenzene does not undergo S N 1 reaction. Reasons: 1. S N 1 is a two-step reaction in which formation of carbocation takes place and the phenyl cation is highly unstable in nature.
Why is chlorobenzene less reactive than chloromethane towards nucleophilic substitution reactions?
Out of chlorobenzene and chloromethane, chloromethane is more reactive towards nucleophilic substitution reactions since it is an alkyl halide. Chlorobenzene is less reactive towards nucleophilic substitution reaction because of the resonance effect and polarity of C − C l bond.
What is a nucleophilic substitution reaction?
Nucleophilic substitution is a fundamental class of reactions in which an electron rich nucleophile selectively bonds with or attacks the positive or partially positive charge of an atom or a group of atoms to replace a so-called leaving group.
Which of the following is most reactive towards nucleophilic substitution chlorobenzene?
Among chlorobenzene and chloroethane, chloroethane is more reactive towards nucleophilic substitution.
What are all the reactions of chlorobenzene?
Chlorobenzene reacts readily with chlorine, nitric acid, or sulfuric acid, forming dichlorobenzenes, chloronitrobenzenes, or chlorobenzenesulfonic acids, respectively, and with chloral in the presence of sulfuric acid to form DDT, an insecticide.
Why benzyl chloride is more reactive than chlorobenzene towards nucleophilic substitution reaction?
Benzyl chloride is more reactive than chlorobenzene towards SN1 reaction because it readily ionises to give benzyl carbonation which is resonance stabilized. In chlorobenzene, ionisation leading to carbonation is not possible. Therefore it is less reactive.
What is the mechanism of reaction of chlorobenzene?
Chlorobenzene reacts with chlorine in presence of FeCl₃ or AlCl₃ to form mixture of o-dichlorobenzene and p-dichlorobenzene. In chlorobenzene,chlorine is deactivating but ortho para directing. During the reaction FeCl₃ or AlCl₃ ,being Lewis acids,abstracts chloride ion from Cl₂ and initiates chloronium ion.
Why is chlorobenzene deactivating?
So, electrophile will attack at Ortho/Para position in chlorobenzene. Cl shows a negative inductive effect due to its high electronegativity means it withdraws the electron density from the benzene ring, so it is a deactivating group.
Why is chlorobenzene O and P directing?
Hint: The ortho/ para directing ability in electrophilic substitution reaction of chlorobenzene is due to the positive resonance effect. In order to solve the question, we must remember that chlorine is ortho and para directing in electrophilic substitution reaction as it is an electron withdrawing group.
Why doesn’t chlorobenzene give a nucleophilic substitution reaction?
In chlorobenzene, the carbon atom to which halogen atom is attached sp2 hybridized and highly electronegative in nature so it cannot be replaced by a nucleophile. Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction.
Does chlorobenzene give SN2 reaction?
Due to resonance, there occurs partial double bond character between carbon and the chlorine . This makes the nucleophilic substitution difficult. Hence Chlorobenzene cannot undergo SN1 or SN2 reaction.
What is an example of a nucleophilic substitution reaction of chlorobenzene?
The halogen of haloarenes can be substituted by OH–, NH2– or CN– with appropriate nucleophilic reagents at high temperature and pressure. Chlorobenzene reacts with ammonium at 250 and at 50 atm to give aniline. Chlorobenzcne reacts with CuCN in presence of pyridine at 250 to give phenyl cyanide.
Why is nucleophilic substitution of para nitro chlorobenzene easier than that of chlorobenzene?
p-chloronitrobenzene undergoes nucleophilic substitution faster than chlorobenzene because p-chloronitrobenzene have nitro group which is electron withdrawing group and chlorine itself is a electronegative group by which chlorine generate electron on its surface and attract as well as it also generate partial positive …
Is chlorobenzene resistant to electrophilic substitution reaction?
Solution. (A) is false, but (R) is true. Explanation: Because of resonance, chlorobenzene acquires a partial double bond, making it extremely reactive to electrophilic substitution processes.
Which is more reactive towards nucleophilic substitution chlorobenzene or para nitro chloro benzene?
Answer: p-nitrochloro benzene is more reactive towards nucleophilic substitution reaction as compared to chlorobenzene. In chlorobenzene, the lone pair on Cl is delocalized with PI electrons of ring. Whereas in p-nitro chloro benzene, presence of strong EWG i.e. NO 2 further delocalizes the negative charge.
Is nucleophilic substitution SN1 or SN2?
SN1 and SN2 are the two forms of nucleophilic substitution reaction. SN1 involves one molecule while Sn2 involves two molecules.
What are the conditions for nucleophilic substitution?
Nucleophilic substitution is the reaction of an electron pair donor (the nucleophile, Nu) with an electron pair acceptor (the electrophile). An sp3-hybridized electrophile must have a leaving group (X) in order for the reaction to take place.
What are the two types of nucleophilic substitution?
It simply represents the Substitution of a Nucleophile for the Leaving Group. There are 2 types of Nucleophilic Substitution Reactions: SN1 & SN2, each having its own mechanism. The SN2 reaction is usually learned first.
Why chlorobenzene is less reactive than ethyl chloride towards nucleophilic substitution reaction?
The lower reactivity of chlorobenzene is due to the partial double bond character of C − C l bond, i.e., it is shorter and stronger bond in comparison to C − C l bond in ethyl chloride. Was this answer helpful?
Why is chlorobenzene less reactive than chlorobenzene?
Chlorobenzene is less reactive than benzyl chloride In chlorobenzene the lone pairs present on Cl atom get involved in resonance with pi electrons of benzene due to which C-Cl bond acquires double bond character Hence reactivity decreases.
Which is more reactive towards sn1 chlorobenzene and benzyl chloride?
Benzyl chloride is highly reactive towards the S N 1 reaction because the intermediate benzyl carbocation formed in the slowest step is stabilized through resonance.
Why is chlorobenzene resistant to nucleophilic substitution reactions?
So, nucleophilic substitution reaction is not possible. Hence, two reasons for less reactivity of chlorobenzene towards nucleophilic substitution reactions are resonance in chlorobenzene and $sp^2$ hybridised nature of carbon atom bonded to chlorine atom.
What are the two chemical reaction of chlorobenzene?
Reaction with NaOH: phenol is obtained by heating chlorobenzene with aqueous NaOH at about 300°C. Reaction with Ammonia: Aniline is obtained by heating chlorobenzene with ammonia at high pressure and around 250°C in the presence of Cu2O.
What is the formation of chlorobenzene reaction?
Chlorobenzene is formed by reaction of chlorine with benzene in the presence of AlCl3. Which of the following species attacks the benzene ring in this reaction? Chlorobenzene is formed by reaction of chlorine with benzene in the presence of A1C13.
Which compounds show nucleophilic substitution reaction?
Acid halides show the least resonance and they form tetrahedral intermediate easily so they are most reactive towards nucleophilic substitution.
What are the reactions of chlorobenzene?
Chlorobenzene undergoes various electrophilic aromatic substation reactions: The nitration of chlorobenzene, where − N O 2 is added in the benzene ring in the presence of nitric and sulfuric acids H N O 3 and H 2 S O 4 .
Why does bromobenzene not undergo nucleophilic substitution?
Answer and Explanation: Bromobenzene is nonreactive under S N 1 conditions as the phenyl cation generated by the dissociation of the bromine atom is highly unstable. And it does not undergo S N 2 reaction as the structure of bromobenzene does not favor the backside attack by the nucleophile.
Why does the nitro group in chlorobenzene increase its reactivity towards a nucleophilic substitution reaction?
Assertion: Presence of a nitro group at ortho or para position increases the reactivity of haloarenes towards nucleophilic substitution. Reason: Nitro group, being an electron withdrawing group decreases the electron density over the benzene ring.
Can chlorobenzene give nucleophilic substitution reactions?
Does chlorobenzene react with NaOH?
Why is chlorobenzene less reactive than alkyl halides?
What are some electrophilic substitution reactions of chlorobenzene?
See more here: What Is The Correct Order Of Nucleophilic Substitution In Chlorobenzene? | Nucleophilic Substitution Reaction Of Chlorobenzene
NUCLEOPHILIC SUBSTITUTION REACTION OF
HALOALKANES AND HALOARENES12 CLASS CHEMISTRY: https://www.youtube.com/playlist?list=PLnIfc62vSK0xwgFqgil6Ykc7cUK2mtN2FD AND F- BLOCK ELEMENTS12 CLASS CHEMIS… YouTube
Nucleophilic Reactions of Benzene Derivatives
An early method of preparing phenol (the Dow process) involved the reaction of chlorobenzene with a concentrated sodium hydroxide solution at Chemistry LibreTexts
Explain the nucleophilic substitution reaction of chlorobenzene.
However, in some adverse conditions and reagents, chlorobenzene can give nucleophilic substitution reactions. – Chlorobenzene when reacted with fused NaOH Vedantu
Nucleophilic Substitution Reaction – Definition, Types,
Nucleophilic substitution reaction is a class of organic reactions where one nucleophile replaces another. It is very similar to the normal displacement reactions which we see in BYJU’S
7.1: Nucleophilic Substitution Reaction Overview
Kinetics of Nucleophilic Substitution Reaction; Let’s start with a simple substitution reaction example: Figure 7.1a Substitution reaction. In this reaction, the Br in the reactant Chemistry LibreTexts
22.4: Preparation of Phenols: Nucleophilic Aromatic
identify the conditions necessary for an aryl halide to undergo nucleophilic aromatic substitution, and give an example of such a reaction. write the detailed mechanism for Chemistry LibreTexts
Explain the electrophilic and nucleophilic substitution reaction of …
(ii) Nucleophilic substitution Reaction of chlorobenzene: Due to resonance, there is a partial double bond character in the C-X bond of chlorobenznene, as a result, the bond Toppr
Explain the nucleophite substitution reaction of chlorobenzene.
Chlorobenzene is less reactive towards nucleophilic substitution reaction because of the following reasons: (1) Resonance effect: The electron pair on chlorine atom is in Toppr
Organic Chemistry I University of Illinois at Chicago UIC
Substitution Reactions Involving Aryl Halides We have not yet seen any nucleophilic substitution reactions of aryl halides. Nucleophilic substitution on chlorobenzene uic.edu
See more new information: activegaliano.org
Nucleophilic Aromatic Substitution Reaction Mechanism – Meisenheimer Complex \U0026 Benzyne Intermediate
Nucleophilic Substitution Reaction Of Chlorobenzene|| Haloalkanes And Haloarenes|12 Class Chemistry
Chlorobenzene Is Extremely Less Reactive Towards A Nucleophilic Substitution Reaction
Nucleophilic Substitution Reaction In Haloarenes Ll Substitution Reaction In Chlorobenzene Neet Jee
Why Haloarenes Are Less Reactive Than Haloalkane Towards Nucleophilic Substitution Reaction ?
Link to this article: nucleophilic substitution reaction of chlorobenzene.
See more articles in the same category here: activegaliano.org/bio