Let’s discuss the question: how many grams of na2o are required to produce 1.60. We summarize all relevant answers in section Q&A of website Activegaliano.org in category: Blog Marketing. See more related questions in the comments below.

How many moles of Na2O are required to produce 160 grams NaOH?
Answer and Explanation: The molar mass of NaOH (sodium hydroxide) is M1=39.997 g/mol M 1 = 39.997 g/mol , and the molar mass of (sodium oxide) Na2O is M2=61.978 g/mol N a 2 O is M 2 = 61.978 g/mol . Thus, the required mass of sodium oxide is equal to 123.9 grams.
How many grams of H2O are required to produce 160 grams NaOH?
Thus, 36 grams of water is needed to produce 160 grams of NaOH.
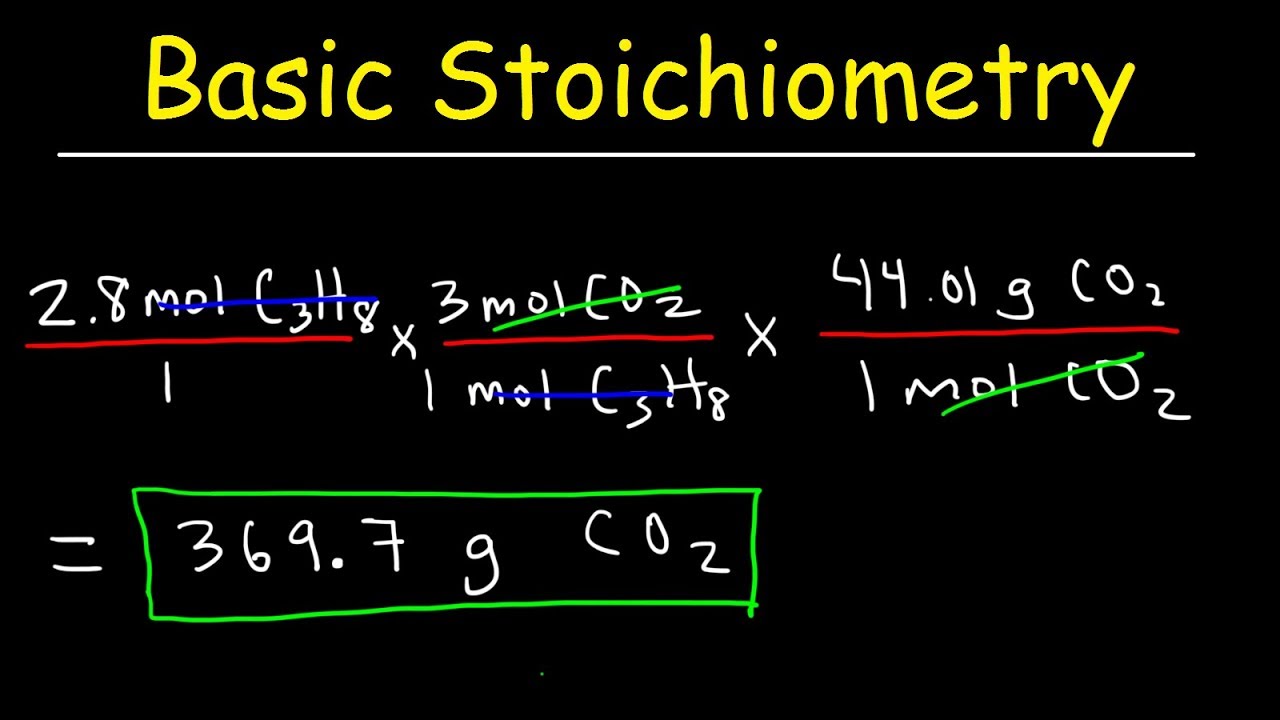
Chemical Reactions (9 of 11) Stoichiometry: Grams to Grams
Images related to the topicChemical Reactions (9 of 11) Stoichiometry: Grams to Grams

How many grams of NaOH is produced from 2 moles of Na2O?
It means 1 moles of Na2 O is required to react with one mol of H2 O to produce 2 moles of NaOH. in terms of mass 1 mole of Na2 O has mass 62 g on reaction with water produces 2 moles of NaOH or 80 g of NaOH. 62 g of Na2 O produces 80 g of NaOH. 124g of Na2 O.
How many grams of NaOH are produced if 120 grams of Na2O react with excess water?
We now need to convert moles to grams. Hence,120 grams of Na2O N a 2 O and excess H2O H 2 O leads to the formation of 155.2 g of NaOH.
How many grams are in a mole of NaOH?
One mole of NaOH has a mass of 40.0 g. If this quantity of NaOH is dissolved in enough water to make exactly 1.00 L of solution, the solution is a 1 M solution.
How many moles of NaOH are produced from 120 grams of Na2O?
Answer. If we begin the problem with 120. grams of Na2O and we are trying to find the mass of NaOH that can be produced, this is a gams to grams stoichiometry problem. The mole ratio from the balanced chemical equation is 2 moles of NaOH for every mole mole of Na2O.
How many grams of sodium sulfate will be formed if you start with 200 grams of sodium hydroxide and you have excess of sulfuric acid?
Therefore, the mass of sodium sulfate formed when we start with 200.0 grams of sodium hydroxide and you have excess sulphuric acid is 355.25g.
What is the molar mass of na2o?
What is the molar mass of NaOH?
How many grams of NaCl are produced when 80.0 grams of O2 are produced?
5gNaCl will be produced with 80.0 grams O2.
How many moles of ammonia could we make from one mole each of N2 and H2?
Using the proportions of the amount of the molecules, we can convert from any reagent or product to any other reagent or product. In the context of this reaction, for every mole of N2 and every 3 moles of H2 we obtain 2 moles of NH3.
How many grams of hydrogen gas are needed to produce 5.00 g of water according to this equation?
Images related to the topicHow many grams of hydrogen gas are needed to produce 5.00 g of water according to this equation?

How many moles of O2 can be produced by letting 12.0 mol of KClO3 react in the following equation?
Terms in this set (10)
How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? The KClO3 / O2 molar ratio is 2/3. 2 mol KClO3 / 3 mol. O2 = 12.00 mol KClO3 / x = 18.00 mol.
How many moles of O2 are formed from 1.65 moles of KClO3?
How many moles of O2 will be formed from 1.65 moles of KClO3? 1.65 mol KClO3 3 mol O2 = 2.475 mol O2 2 mol KClO3 2.
How many grams of hydrogen are produced if 120g of sodium are available?
120 g of sodium will result in the formation of 5.21 g of hydrogen gas.
How are chemical equation balanced?
If each side of the equation has the same number of atoms of a given element, that element is balanced. If all elements are balanced, the equation is balanced.
How many moles are there in 80g of sodium hydroxide?
= 80 / 40= 2 mol .
What is the formula for moles to grams?
Multiply the given number of moles (2.50 mol) by the molar mass (122.548 g/mol) to get the grams.
What is the mole of nh3?
17.03052 g/mol .
What is the mass in grams of 1.5 moles of sodium chloride?
1.5 Moles of Sodium = 34.484655 Grams
Please enter another number of moles of sodium that you want converted to grams.
How many chlorine molecules are there in 71 g of gaseous chlorine?
The correct answer is 1.205 × 1024. Weight of Cl2 =71 g. The number of moles of Cl2= 71 g/71g/gatom=1 moles. Avogadro’s number is 6.023×1023 molecules/mol.
How many grams of sodium sulfate will be formed if you start with 1.25 l of a 4.00 M solution of sodium hydroxide?
Hence 360 grams of Na2SO4 will be formed from the given 1.25 L of a 4.0 M solution of sodium hydroxide.
Stoichiometry Basic Introduction, Mole to Mole, Grams to Grams, Mole Ratio Practice Problems
Images related to the topicStoichiometry Basic Introduction, Mole to Mole, Grams to Grams, Mole Ratio Practice Problems
What is the formula of sodium sulfate?
How many grams of lino3 will be needed to make 250 grams li2so4?
The ratio is 4:2, so we will need 4 moles of Lithium Nitrate to react completely with Lead IV Sulfate to produce 2 moles of Lithium Sulfate. 1 mole of Lithium Nitrate is 68.951 g/mol. 1 mole of Lithium Sulfate is 103.11 g/mol. ANSWER: 93.3 grams Lithium Nitrate are needed.
Related searches
- how many grams of na2o are required to produce 1.60
Information related to the topic how many grams of na2o are required to produce 1.60
Here are the search results of the thread how many grams of na2o are required to produce 1.60 from Bing. You can read more if you want.
You have just come across an article on the topic how many grams of na2o are required to produce 1.60. If you found this article useful, please share it. Thank you very much.