Let’s discuss the question: how many molecules are in 5.0 mg of aspartame. We summarize all relevant answers in section Q&A of website Activegaliano.org in category: Blog Marketing. See more related questions in the comments below.

How many molecules is 5 grams?
01 +15.99. Therefore molecular mass of water (M) is equal to 18,01. So in 5g sample of water there is 1.67 x 1023 water molecules.
What is the mass in grams of 1 molecule of aspartame?
Calculate the molar mass of aspartame. 294.3 g/mol b.
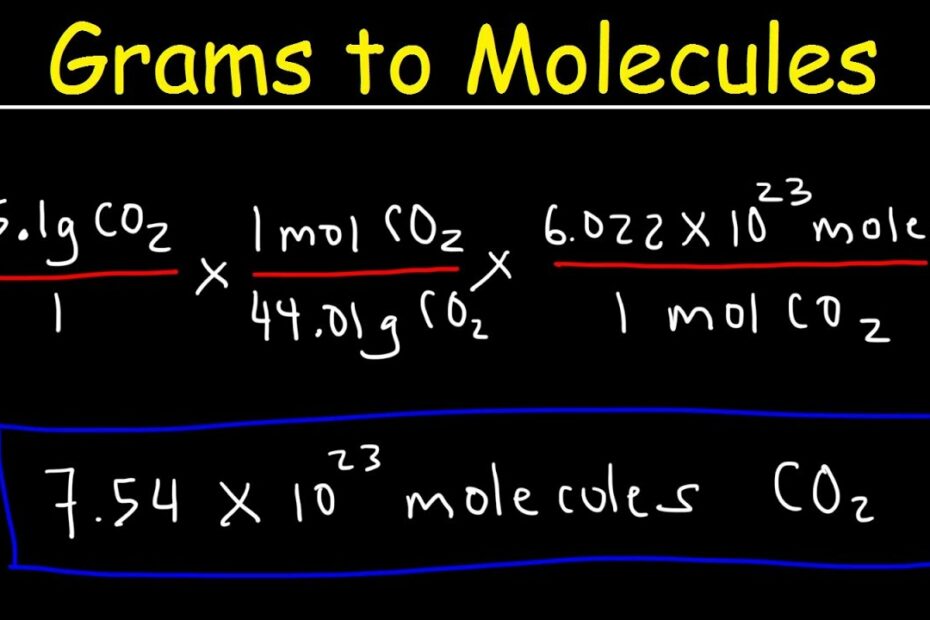
Grams to Molecules and Molecules to Grams Conversion
Images related to the topicGrams to Molecules and Molecules to Grams Conversion

What is the number of moles of aspartame?
We can do this using the molar mass of the substance and Avogadro’s constant, 6.022×1023 . Aspartame has the chemical formula C14H18N2O5 , and its molar mass is calculated to be 294.30 g/mol .
How many moles of aspartame are present in 225g of aspartame?
How many moles of aspartame are present in 225 g of aspartame. =0.765 moles.
How do you calculate molecules?
Explanation: Determine the mass of the substance, and its molar mass. Divide the given mass by its molar mass to get moles, then multiply times 6.022×1023molecules1mol .
How many molecules are in 25g of nh3?
1 Expert Answer
molecules NH3 = 1.47 moles x 6.02×1023 molecules/mole = 8.9×1023 molecules of NH3 (to 2 sig. figs.)
What is the percent composition of aspartame?
C=57.14%H=6.16%N=9.52%O=27.18%
What is the mass of 5 moles of Fe2O3?
What is the mass of 5 moles of Fe2O3 ? Smal x 159.0 1798.45g Page 2 3.
How do you find the mass of one mole of aspartame?
The molar mass of a compound can be calculated by adding the standard atomic masses (in g/mol) of the constituent atoms. How many moles are in 5 mg of aspartame? Mass of aspartame = 5 mg = 0.005 grams. Also, molar mass of aspartame = 294.3 grams.
What is the chemical formula for aspartame?
How many molecules are in 0.400 moles of N2O5?
1 Answer. 0.400 mol N2O5 contains 2.41 molecules N2O5 .
How do you convert moles to molecules?
- To go from moles to molecules, multiply the number of moles by 6.02 x 1023.
- To go from molecules to moles, divide the numbers of molecules by 6.02 x 1023.
Calculate the Mass of a Single Atom or Molecule
Images related to the topicCalculate the Mass of a Single Atom or Molecule
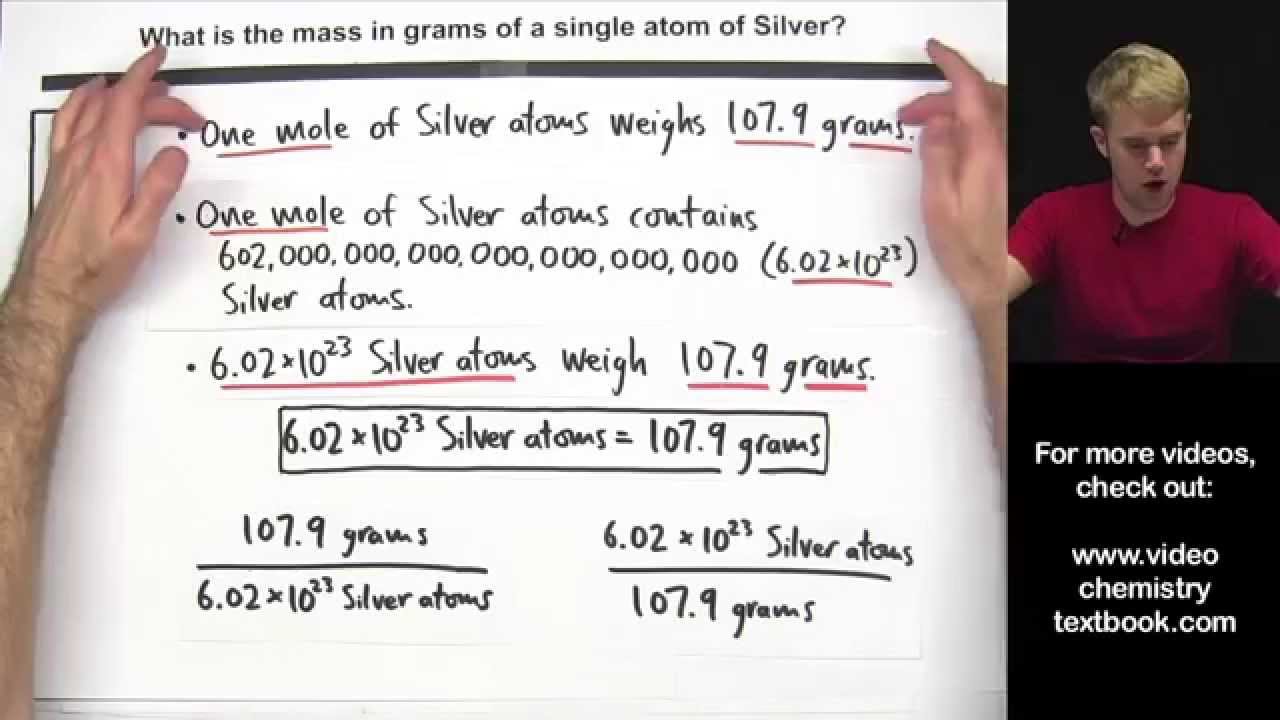
How many grams are in 4.5 moles of CO2?
The mass of 4.5 moles of CO2 is 198.
What is the mass of one mole of aspartame having formula c14h18n2o5?
Thus, molar mass of aspartame = 294 g.
What is the mass of one molecule of neotame?
| Property Name | Property Value |
|---|---|
| Exact Mass | 378.21547206 |
| Monoisotopic Mass | 378.21547206 |
| Topological Polar Surface Area | 105 Ų |
| Heavy Atom Count | 27 |
How many molecules are in a cell?
Ho’s analysis reveals for the first time how many molecules of each protein there are in the cell, with a total number of molecules estimated to be around 42 million. The majority of proteins exist within a narrow range — between 1000 and 10,000 molecules.
What is number of molecules in 9.0 g of steam?
9 gram of steam contain 3.015*10^23 H2O molecules. Note: 9 gram of water whether as ice or liquid water or steam always contains 3.015*10^23 water molecules.
How many molecules are in 1.5 moles?
Hence, number of molecules in 1.5 moles of ammonia is 9.033 × 1023.
What is the percent composition of potassium in KCN?
| Element | Symbol | Mass Percent |
|---|---|---|
| Kalium | K | 60.045% |
| Carboneum | C | 18.446% |
| Nitrogenium | N | 21.511% |
How many molecules of ammonia NH3 are produced?
We see that 1 molecule of nitrogen reacts with 3 molecules of hydrogen to form 2 molecules of ammonia.
How many grams are in 5 moles of NH3?
Therefore, the mass of 5 mole of ammonia is 85 gm.
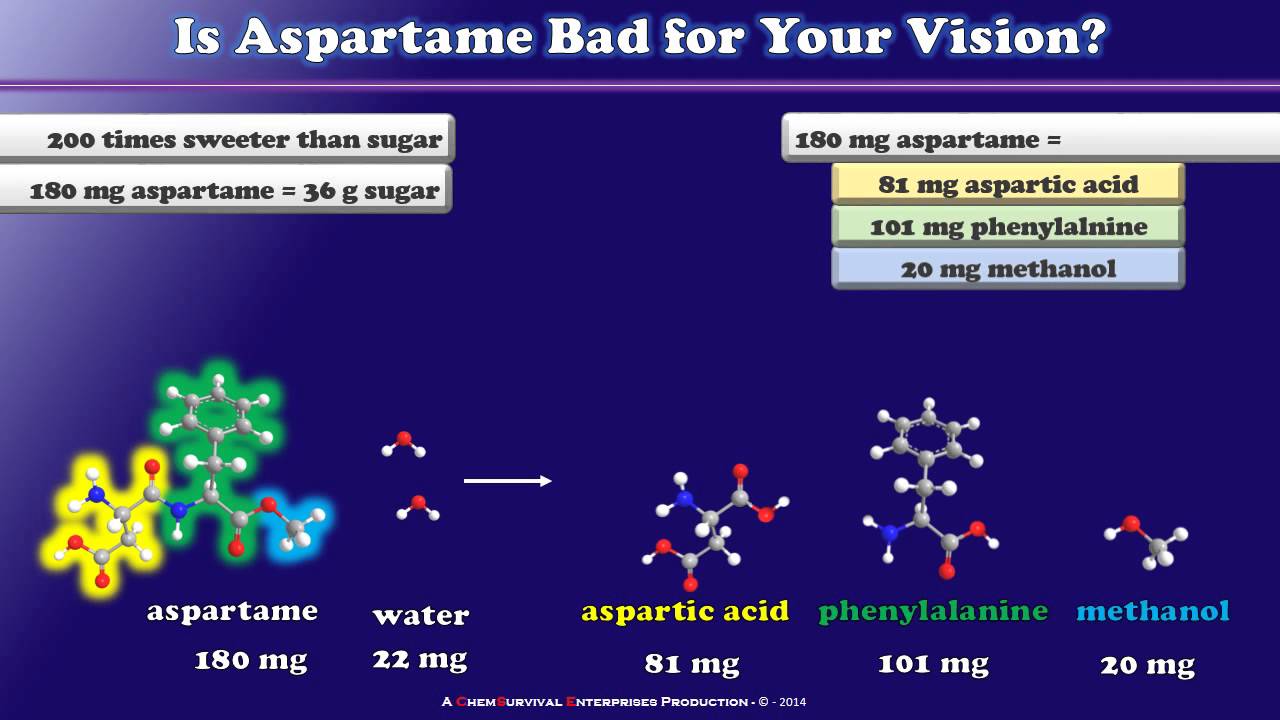
What is the composition of each of the components of aspartame?
Aspartame is a methyl ester of a dipeptide consisting of two amino acids, aspartic acid, and phenylalanine (Figure 1). In the digestive tract, aspartame is completely hydrolyzed to its constituent amino acids in addition to methanol.
The Chemistry of Aspartame
Images related to the topicThe Chemistry of Aspartame
How do you find the percent composition?
- Find the molar mass of all the elements in the compound in grams per mole.
- Find the molecular mass of the entire compound.
- Divide the component’s molar mass by the entire molecular mass.
- You will now have a number between 0 and 1. Multiply it by 100% to get percent composition.
What is the number of atoms in aspartame?
| Property Name | Property Value | Reference |
|---|---|---|
| Heavy Atom Count | 21 | Computed by PubChem |
| Formal Charge | 0 | Computed by PubChem |
| Complexity | 380 | Computed by Cactvs 3.4.8.18 (PubChem release 2021.05.07) |
| Isotope Atom Count | 0 | Computed by PubChem |
Related searches
- how many hydrogen atoms are present in 1 50 mg of aspartame
- how many atoms are in 1 2 g of aspartame
- how many atoms are in 1.2 g of aspartame.
- how many hydrogen atoms are present in 4 50 mg of aspartame
- how many molecules are in 100g of aspartame
- how many hydrogen atoms are present in 4.50 mg of aspartame?
- how many hydrogen atoms are in 1 mg of aspartame
- how many hydrogen atoms are present in 5 00 mg of aspartame
- how many atoms of nitrogen are in 1 2000 grams of aspartame
- how many hydrogen atoms are present in 1.50 mg of aspartame?
- how many molecules are in 100g of aspartame?
- how many atoms of nitrogen are in 1.2 grams of aspartame
- how many atoms of nitrogen are in 1 2 grams of aspartame
Information related to the topic how many molecules are in 5.0 mg of aspartame
Here are the search results of the thread how many molecules are in 5.0 mg of aspartame from Bing. You can read more if you want.
You have just come across an article on the topic how many molecules are in 5.0 mg of aspartame. If you found this article useful, please share it. Thank you very much.