Let’s discuss the question: how many moles of kclo3 must be reacted. We summarize all relevant answers in section Q&A of website Activegaliano.org in category: Blog Marketing. See more related questions in the comments below.

How many moles are in KClO3?
How many grams KClO3 in 1 mol? The answer is 122.5495. We assume you are converting between grams KClO3 and mole.
What is the mole ratio of KClO3 to o2 in this reaction?
So, our mole ratio of potassium chlorate to oxygen is 2:3 .
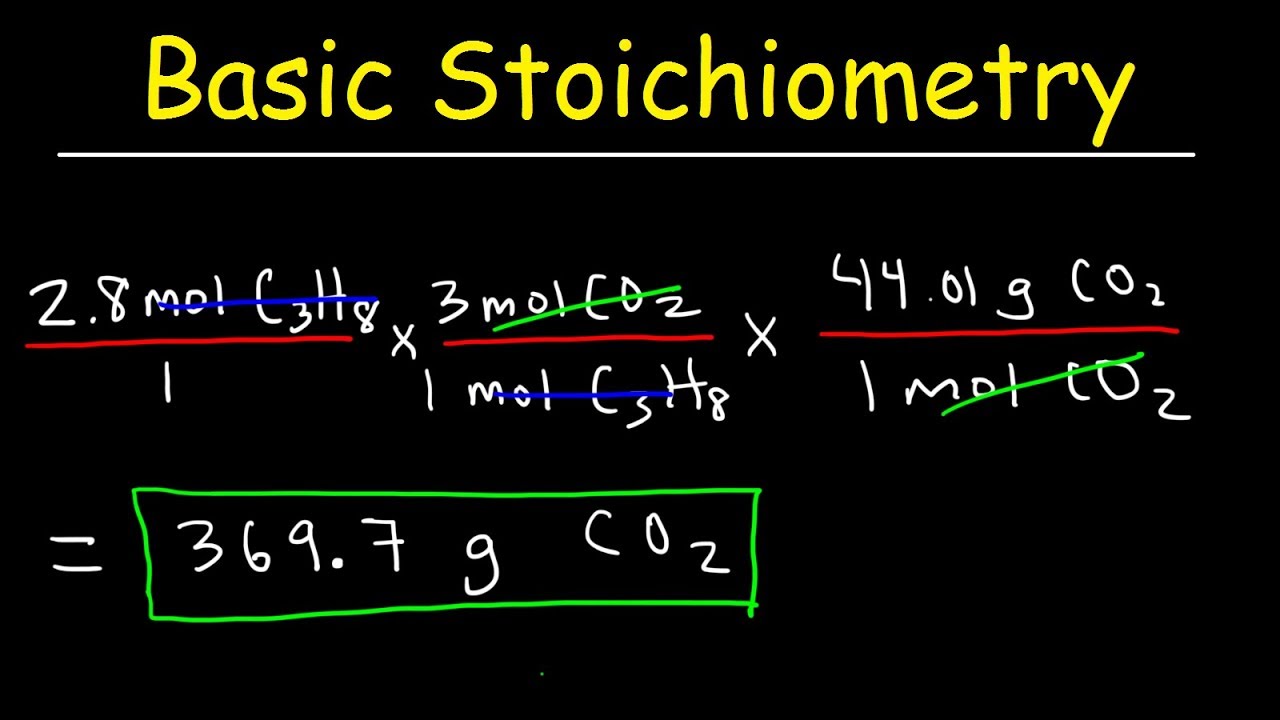
Stoichiometry Mole to Mole Conversions – Molar Ratio Practice Problems
Images related to the topicStoichiometry Mole to Mole Conversions – Molar Ratio Practice Problems

How many moles of O2 are produced when 4 moles of KCl are produced?
Therefore, 4 moles of potassium chlorate must be used to produce 6 moles of oxygen gas.
How many moles of KClO3 give 3 moles of oxygen a 2 mole B 1 moles C 4 mole d 5 moles?
Thus, 3 moles of oxygen will be supplied by 2 moles of KClO3.
What is the total number of moles of KClO3 is needed to produce 6 moles of O2 g?
∴ 4 moles of KClO3 will produce 6 moles of oxygen.
How do I calculate moles?
- Measure the weight of your substance.
- Use a periodic table to find its atomic or molecular mass.
- Divide the weight by the atomic or molecular mass.
- Check your results with Omni Calculator.
What is the mole mole ratio fe2o3?
Explanation: Yes, the mole ratio that exists between iron and iron(III) oxide can be written as 4:2 , i.e. 2 moles of iron(III) oxide require 4 moles of iron.
How many moles of O2 can be produced from 12 moles KClO3 react?
How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? The KClO3 / O2 molar ratio is 2/3. 2 mol KClO3 / 3 mol. O2 = 12.00 mol KClO3 / x = 18.00 mol.
What is the mole mole ratio of NH3 nso4 2SO4?
Answer: The balanced equation indicates that 2 moles of NH3 react to produce 1 mole of (NH4)2SO4. Because the reaction occurs at STP, 1 mole of NH3 gas occupies 22.4 L.
How many moles of KClO3 are required to produce 2.4 moles O2?
x=1.6moles
Therefore 1.6 moles of KClO3 are required to produce 2.4 moles of O2.
How many moles of oxygen are produced when 3 moles of KClO3 decomposes?
2KClO3 –> 2KCl + 3O2
(3/2)*(3.0) moles O2, or 4.5 moles of O2. Kaitlyn G.
Which of the following will be correct when 4 moles of KClO3?
Hence, 4 moles of KClO3 are required to give 6 moles of oxygen.
Mole Ratio Practice Problems
Images related to the topicMole Ratio Practice Problems

How many moles of hydrogen will be needed to react with 2 moles of nitrogen according to the following?
Expert-verified answer
To thoroughly react 2 moles of N2, 6 moles of H2 gas are required.
How many moles of oxygen are consumed of 8 moles H2 are used?
4 moles of O2 will be consumed from 8 moles of H2 when the two substances react to form water.
How many moles of water are produced when 5 moles of oxygen are consumed?
How many moles of water are produced when 5.00 moles of oxygen are used? Answer: 10.0 mol of water (H2O) are produced.
How many moles of carbon dioxide are produced when 5.0 moles of butane react completely?
| Questions | Answer |
|---|---|
| 36 Given the balanced equation for the reaction between butane and oxygen: 2C4H10 + 13O2 –> 8CO2 + 10 H2O + Energy How many moles of carbon dioxide are produced when 5.0 moles of butane react completely? (1) 5.0 mol (2) 10. mol (3) 20. mol (4) 40. mol | 3 |
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate?
Explanation: From the equation, 32 equiv dixoygen gas evolve from 1 equiv chlorate. You started with 6 equiv chlorate, and thus 9 equiv dioxygen gas are evolved.
How many atoms are in 3.75 moles?
Answer and Explanation: There are 2.26 x 1024 carbon atoms in 3.75 moles of carbon. A mole is defined as 6.022 x 1023 elementary entities of a… See full answer below.
What is a 1 mole?
A mole is defined as 6.02214076 × 1023 of some chemical unit, be it atoms, molecules, ions, or others. The mole is a convenient unit to use because of the great number of atoms, molecules, or others in any substance.
What does n m/m mean?
m = mass of the pure substance (measured in grams, g) n = amount of the pure substance (measured in moles, mol) ⚛ This mathematical equation can be rearranged to give the following: (i) n = m ÷ M. moles = mass ÷ molar mass.
How do you find moles from molarity?
- Find the molarity and volume of your solution.
- Make sure that the units for the volume are the same as for the volume part of the molarity (e.g., mL and mol/mL).
- Multiply the volume by the molarity. This is the number of moles present.
What is a mole to mole ratio?
The mole ratio is the ratio of moles of one substance to the moles of another substance in a balanced equation. Use of mole ratios allows us to convert from one chemical substance to another.
Stoichiometry Basic Introduction, Mole to Mole, Grams to Grams, Mole Ratio Practice Problems
Images related to the topicStoichiometry Basic Introduction, Mole to Mole, Grams to Grams, Mole Ratio Practice Problems
What is the mole ratio in Fe o2 → Fe2O3?
In this reaction, four moles of Fe reacts with three moles of oxygen to form two moles of ferric oxide. Thus, the mole ratio of Fe and ferric oxide is 4/2.
What is the mole ratio of iron and oxygen?
The Law of Multiple Proportions
The ratio of mass of oxygen to iron is 0.2865. It’s not a ratio of two integers, but the ratio is always the same, regardless of how much iron and oxygen there is. Now iron and oxygen also form a different compound called “ferric oxide”.
Related searches
- how many moles of h are required to give off 2251 kj of heat in the following reaction
- how many moles of kclo3 are needed to make 3.50 moles of kci
- how many moles of kclo3 must completely react to produce 6 moles of o2
- what is the change in enthalpy in kilojoules when 2.30 mol of mg
- how many moles of h₂ are required to give off 2251 kj of heat in the following reaction
- what is the change in enthalpy in kilojoules when 3 24 g of ch3oh
- how many moles of kclo3 must completely react
- how many grams of c6h6 liquid must decompose
- how many moles of potassium chlorate
- what is the mole mole ratio of kclo3 to o2
- how many moles of kclo3
- what is the change in enthalpy in kilojoules when 2 30 mol of mg
- what is the number of moles of c that must completely
Information related to the topic how many moles of kclo3 must be reacted
Here are the search results of the thread how many moles of kclo3 must be reacted from Bing. You can read more if you want.
You have just come across an article on the topic how many moles of kclo3 must be reacted. If you found this article useful, please share it. Thank you very much.
