Let’s discuss the question: how many sigma bonds are present in the following compound. We summarize all relevant answers in section Q&A of website Activegaliano.org in category: Blog Marketing. See more related questions in the comments below.
How many sigma bonds are present in the given compound?
Therefore, total number of sigma bond is 18. Was this answer helpful?
How many sigma and pi bonds are present in the following compound ch3 CH2 CH CH COOH?
Answer: There are seven sigma bonds and three pi bonds in CH2=CH-CH=CH2. From left till right the carbons are one, two, three, and four: 3H are attached to 1C, so there are three sigma bonds. In C2 one sigma bond and one pi bond are present.
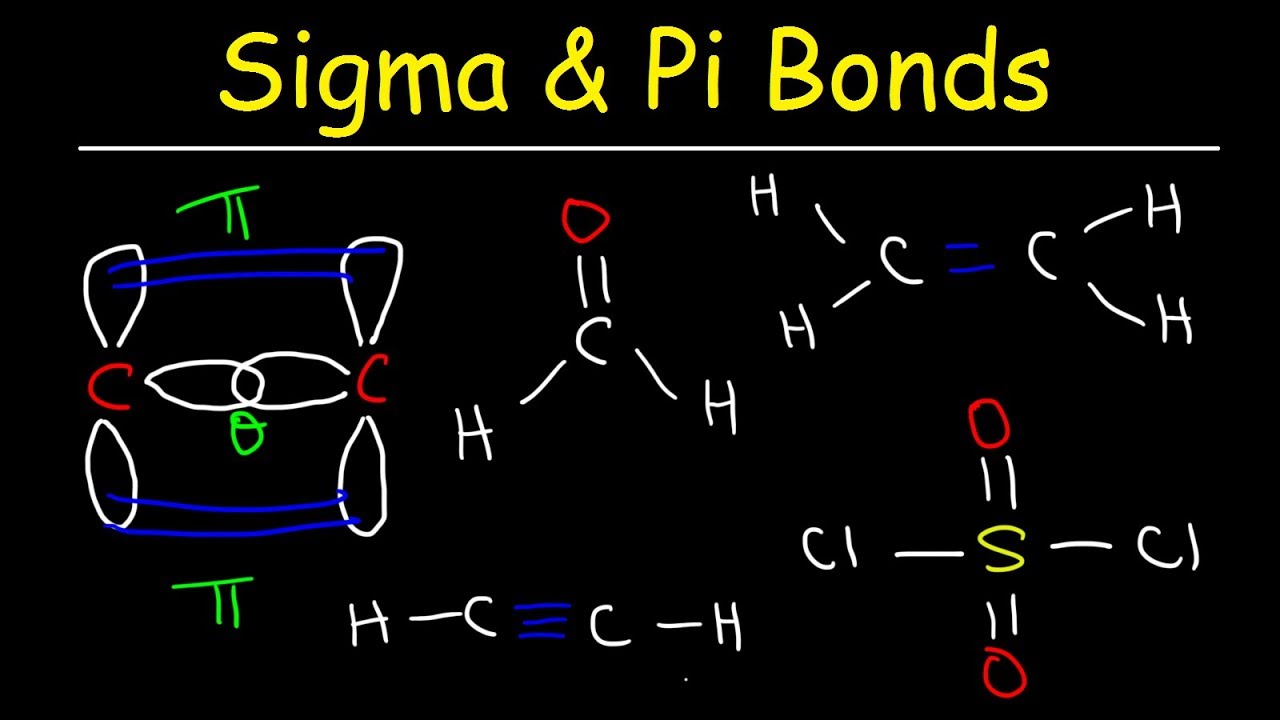
Sigma and Pi Bonds Explained, Basic Introduction, Chemistry
Images related to the topicSigma and Pi Bonds Explained, Basic Introduction, Chemistry

How do you count the sigma bonds?
…
1 Answer
- Single bond = 1 sigma bond.
- Double bond = 1 sigma and one pi bond.
- Triple bond = 1 sigma and 2 pi bonds.
How many sigma and pi bonds are present in c2h2 and c2h4?
Hence the answer for a) is 3,2 and b)5,1.
How many sigma bonds are present in p4o10?
The number of sigma bonds (σ-bonds) in P4O10 is 16.
How many pi bonds are present in compound?
Each double bond contains one sigma and one pi bond. There are two double bonds. Thus, there are 2 pi bonds.
How many sigma bonds and pi bonds are present?
Every single bond is a sigma bond and every multiple bond contains one sigma bond. Thus, it has 8 sigma bonds and 4 pi bonds.
How many sigma bonds and pi bonds are present in the following compound ch3ch CHCN?
There are 9 sigma bonds and 3 pi bonds present in .
How many sigma bonds and pi bonds are present in ch2 ch2?
In total there are nine sigma and two pi bonds are present.
How many sigma bonds are present in acetylsalicylic acid?
There are 21 sigma bonds.
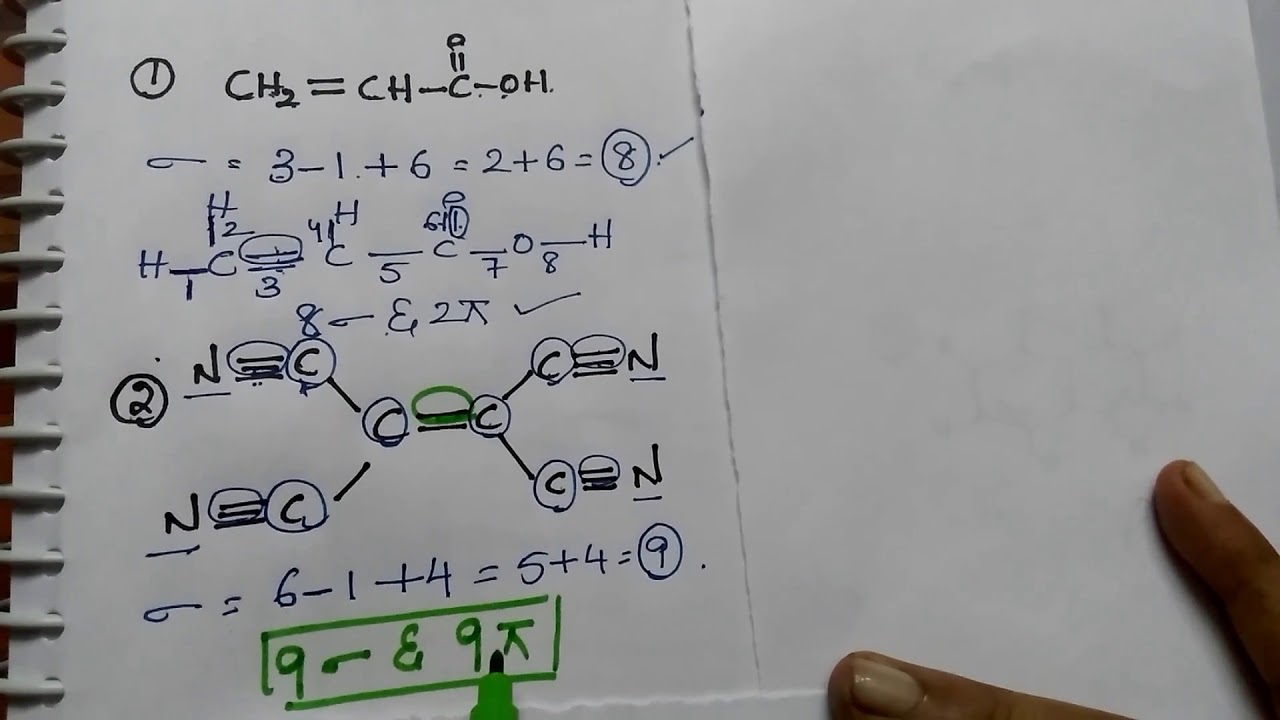
Trick To Find Number Of Sigma and Pi Bonds
Images related to the topicTrick To Find Number Of Sigma and Pi Bonds
How many sigma bonds are in benzene?
As in structure, There are 6 C-H & 6 C-C bonds. Therefore, it is 12 sigma bonds. There are 3 C=C pi bonds in benzene.
How many sigma bonds are in nf3?
Nitrogen trifluoride (NF3) lewis structure contains three sigma bonds and one lone pair around nitrogen atom.
How many sigma bonds are in C2H4?
5 sigma and one pi bond.
How many bonds does C2H4 have?
C2H4 Lewis structure contains four C-H bonds and one double bond in between two carbon atoms. No lone pair is present on the central or outer atom in the lewis structure of C2H4.
How many sigma bonds are there in ch4?
Methane molecule contains 4 σ and 0 π bonds.
How many pop bonds are present in P4O10?
P4O10 molecule contains six POP bonds.
How many sigma and pi bonds are there in toluene?
Therefore, toluene has 15 sigma bonds and 3pi bonds.
How do you find sigma and pi bonds in a compound?
Open Chain Olefinic Hydrocarbons
E.g.: In C176H250, X = 176, Y = 250, therefore P = (2 x 176 – 250)/2 +1 = 51 + 1 = 52 number of π bonds or double bonds. where, X = number of carbon atoms; Y = number of hydrogen atoms and S = number of sigma bonds (σ-bonds).
How many sigma bonds are in a triple bond?
Single bonds consist of one sigma (σ) bond, double bonds have one σ and one pi (π) bond, and triple bonds have one σ and two π bonds.
How Many Sigma and Pi Bond (Count Number of Sigma and Pi Bonds) Example, Practice Problem, Shortcut
Images related to the topicHow Many Sigma and Pi Bond (Count Number of Sigma and Pi Bonds) Example, Practice Problem, Shortcut
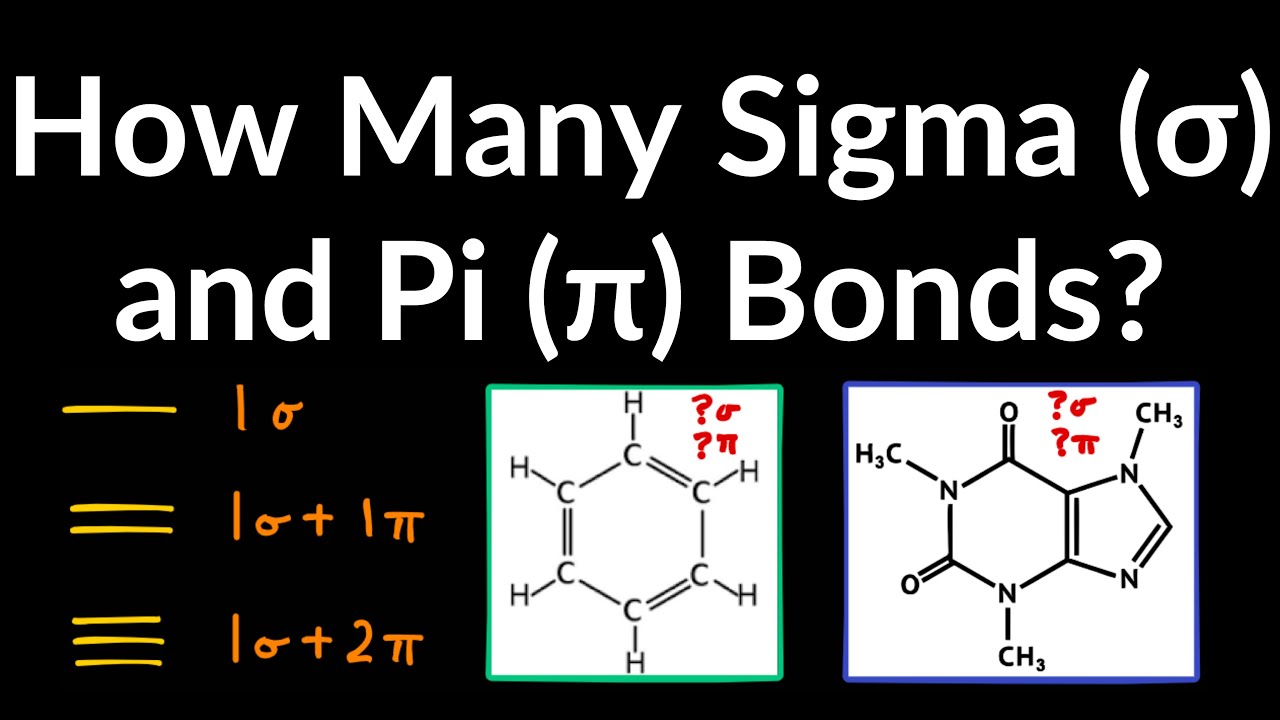
How many pi and sigma bonds are present in benzene?
Nine sigma and three pi.
How many sigma bonds are there in CH3 CH ch2?
Solution : Eight `sigma ` and one `pi` bonds.
Related searches
- how many sigma and pi bonds are present in the following compound
- how many sigma and pi bonds are present in acetylene
- how many sigma and pi bonds are present in ethene
- how many sigma and pi bonds are present in benzene
- how many sigma and pi bonds are in the following compounds
- how many sigma and pi bonds are present in each of the following molecules
- how many sigma and pi bonds are present in toluene
- q 1.18 how many sigma bonds are present in the following compound
- how many sigma bonds are present in the given compound
- how many σ and π bonds are in each compound? co
- how to find the number of sigma bonds in a molecule
- how many sigma and pi bonds are present in ethyne
- how to tell how many sigma bonds are in a molecule
- how many sigma bonds are present in the final product (organic compound) in the following reaction
- how many sigma and pi bonds are present in
- how many σ (sigma) and π (pi) bonds are present in each of the following molecules
- how many and bonds are in each compound co
Information related to the topic how many sigma bonds are present in the following compound
Here are the search results of the thread how many sigma bonds are present in the following compound from Bing. You can read more if you want.
You have just come across an article on the topic how many sigma bonds are present in the following compound. If you found this article useful, please share it. Thank you very much.