Let’s discuss the question: write a balanced equation showing the decomposition of carbonic acid.. We summarize all relevant answers in section Q&A of website Activegaliano.org in category: Blog Marketing. See more related questions in the comments below.
How does carbonic acid decompose?
Carbonic acid, H2CO3, is stable at ambient temperatures in strictly anhydrous conditions. It decomposes to form carbon dioxide in the presence of any water molecules. Carbonic acid forms as a by-product of CO2/H2O irradiation, in addition to carbon monoxide and radical species (HCO and CO3).
What type of reaction is decomposition of carbonic acid?
Carbonic acid (H2CO3) is an ingredient in soft drinks. A decomposition reaction takes place when carbonic acid breaks down to produce water (H2O) and carbon dioxide (CO2). This occurs when you open a can of soft drink and some of the carbon dioxide fizzes out.
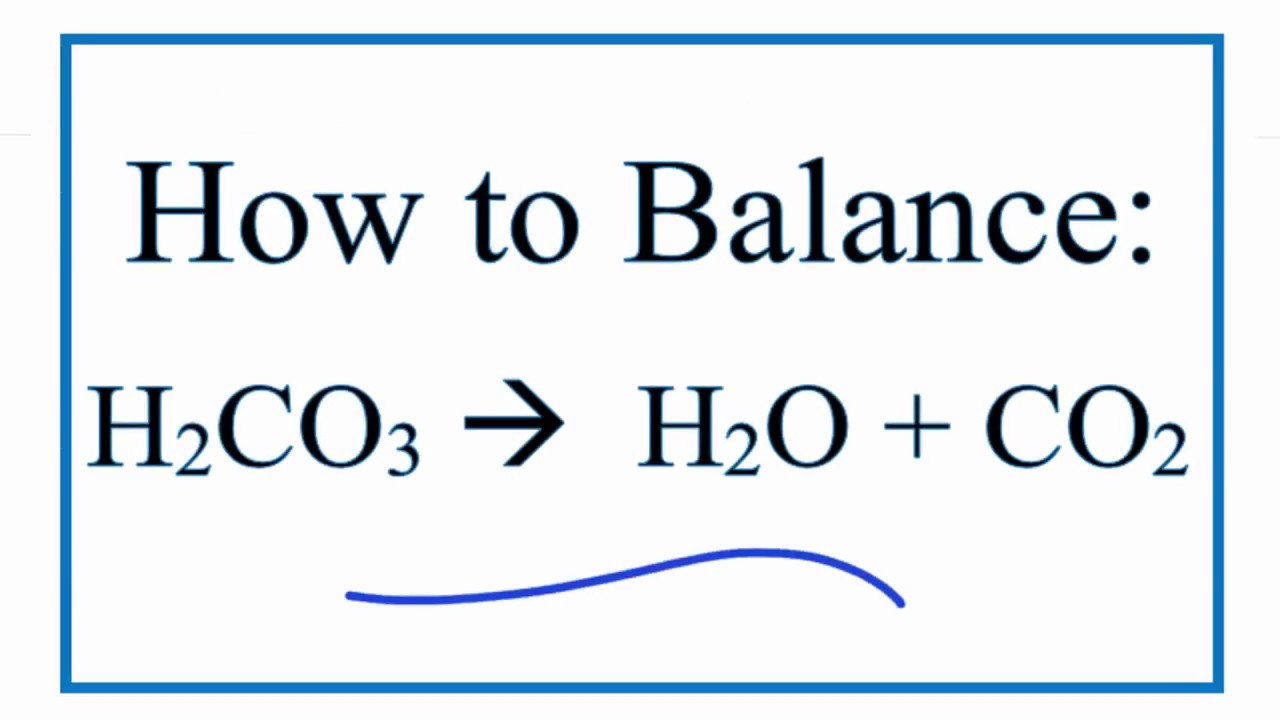
How to Balance H2CO3 = H2O + CO2 (Decomposition of Carbonic acid)
Images related to the topicHow to Balance H2CO3 = H2O + CO2 (Decomposition of Carbonic acid)

What is the equation for carbonic acid?
What is the photosynthesis equation?
The process of photosynthesis is commonly written as: 6CO2 + 6H2O → C6H12O6 + 6O2. This means that the reactants, six carbon dioxide molecules and six water molecules, are converted by light energy captured by chlorophyll (implied by the arrow) into a sugar molecule and six oxygen molecules, the products.
Is the decomposition of carbonic acid spontaneous?
Commonly seen as the “fizz” in carbonated beverages, carbonic acid will spontaneously decompose over time into carbon dioxide and water. The reaction is written as: H2CO3 → H2O + CO. Other carbonates will decompose when heated to produce their corresponding metal oxide and carbon dioxide.
How long does it take for carbonic acid to decompose?
Absolutely pure carbonic acid does not spontaneously decompose. In the absence of water, the half-life for the decomposition is 180 000 years.
What is the decomposition of sulfuric acid?
Sulfuric acid is decomposed to SO2 at high temperature, and after heat recovery in a boiler, the exhaust is quenched with water and purified to yield pure sulfur dioxide. A catalytic converter is then used to convert SO2 to SO3 which is the starting material for the production of pure sulfuric acid and oleum.
Which is the balanced chemical equation showing hydrogen peroxide decomposing into hydrogen and oxygen?
h2o2 → h2 + o2 4.
How do you balance a decomposition reaction?
You balance decomposition reactions just like any other reaction. Add coefficients in front of each formula so that the number of atoms of each element are the same on both sides, being careful not to change formulas by changing subscripts.
What are 3 examples of decomposition reaction?
- Production of calcium oxide or quicklime.
- Production of lithium oxide.
- Preparation of oxygen and carbon dioxide.
- In metallurgy, for the extraction of metals from their oxides and chlorides through electrolytic decomposition.
H2CO3=CO2+H2O Balanced Equation|Carbonic acid =Carbon dioxide and Water Balanced Equation
Images related to the topicH2CO3=CO2+H2O Balanced Equation|Carbonic acid =Carbon dioxide and Water Balanced Equation

What is the formula for hydrobromic acid?
What has carbonic acid?
Carbonic acid is present in blood in the human body. It is formed in the human body when water gets dissolved with carbon dioxide. It is also present in rainwater, calcite, fermentation, coal, groundwater, meteors, volcanoes, amino acids, proteins, oceans, plants, erythrocytes, sulphur deposits, salts, and caves.
What is the correct formula for perchloric acid?
What is the balanced equation for CaCO3 CaO CO2?
Search by reactants (CaCO 3) and by products (CaO, CO 2)
| 1 | CaCO3 → CO2 + CaO |
|---|---|
| 4 | Ca(OH)2 + CaCO3 → H2O + CO2 + CaO |
What is the balanced symbol equation for photosynthesis?
The chemical equation for photosynthesis is 6CO2+6H2O→C6H12O6+6O2.
How does photosynthesis occur give the balanced chemical equation for photosynthesis?
6CO2 + 6H2O — > C6H12O6 + 6O2. Generally, photosynthesis utilizes light vitality to change over carbon dioxide into a starch.
What is photosynthesis class7?
Photosynthesis. Photosynthesis. The process by which plants make their own food in the presence of sunlight, carbon-dioxide present in air, water, minerals and chlorophyll present in leaves is termed as photosynthesis.
What is decomposition in Chem?
Definition. Decomposition is the breakdown or change of a material or substance (by heat, chemical reaction, or other processes) into other chemical compounds. Thermal decomposition is decomposition initiated by heating.
Carbonic Acid-Bicarbonate Equation \u0026 pH homeostasis
Images related to the topicCarbonic Acid-Bicarbonate Equation \u0026 pH homeostasis

What is the process of decomposition?
Decomposition is the first stage in the recycling of nutrients that have been used by an organism (plant or animal) to build its body. It is the process whereby the dead tissues break down and are converted into simpler organic forms.
What type of chemical reaction is decomposition?
A decomposition reaction is a type of chemical reaction in which one reactant yields two or more products. Decomposition reactions are also known as analysis reactions or chemical breakdowns. The opposite of this type of reaction is a synthesis, in which simpler reactants combine to form a more complex product.
Related searches
- h2co3 h2o co2
- write the chemical equation for the reaction of carbonic acid h2co3 with water
- h2co3 h2o reaction
- carbonic acid formula
- h2co3 balanced equation
- decomposition of carbonic acid equation
- carbonic acid decomposition reaction
- carbonic acid is heated balanced equation
- h2co3 + h2o reaction
Information related to the topic write a balanced equation showing the decomposition of carbonic acid.
Here are the search results of the thread write a balanced equation showing the decomposition of carbonic acid. from Bing. You can read more if you want.
You have just come across an article on the topic write a balanced equation showing the decomposition of carbonic acid.. If you found this article useful, please share it. Thank you very much.
